Hydrogen and its compoundHard
Question
Compound X of molecular formula C4H6 takes up one equivalent of hydrogen in presence of Pt to form another compound Y which on ozonolysis gives only ethanic acid. The compound X can be
Options
A.CH2 = CH - CH = CH2
B.CH2 = C = CHCH3
C.CH3C = CCH3
D.All the three
Solution
Formation of only CH3COOH by ozonolysis indicates that the compound Yshould be CH3CH = CHCH3 which can be formed by all of the the three given compounds
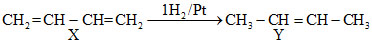
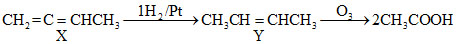
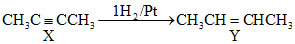
Create a free account to view solution
View Solution FreeMore Hydrogen and its compound Questions
The types of bonds present in CuSo4. 5H2O are only :...(a) H2O2 + O3 → H2O + 2O2 (b) H2O2 + Ag2O → 2Ag + H2O + O2 Role of hydrogen peroxide in the above reactions ...Potassium manganate (K2 MnO4) is formed when :...Which one of the following produces acyl halide by treatment with PCl5...1-Buttyne and cold alkaline KMnO4 react to produce :...