Coordination CompoundHard
Question
[Fe(NO2)3Cl3] and [Fe(ONO)3Cl3] shows
Options
A.linkage isomerism
B.geometrical isomerism
C.optical isomerism
D.none of the above
Solution
[Cu(The given compound contains - NO2 group which can donate electrons either from O- or from N-. Thus, it shows linkage isomerism. Thus the two give compounds are linkage isomers, pone is nitrite - O - N = O fromamd the other is nitro, 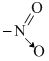 from.NH3)4]2+
from.NH3)4]2+
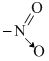 from.NH3)4]2+
from.NH3)4]2+Create a free account to view solution
View Solution FreeMore Coordination Compound Questions
Which of the following are square planar complexes ?(1) [AuCl4]- (2) [Pt(Cl)4]2- (3) [Mn(Br)4]2- (4) [Cu(NH3)4]2+Select ...Which one of the following species does not represent cationic species of vanadium formed in aqueous solution...Among TiF62–, CoF63– , Cu2Cl2 and – NiCl42– the colourless species are:...π-bonding is not involved in:...The magnetic moment of [NiX4]2– ion is found to be zero. Then the metal of the complex ion is (X = monodentate anionic l...