Photoelectric EffectHard
Question
A 200 W sodium street lamp emits yellow light of wavelength 0.6 μm. Assuming it to be 25% efficientin converting electrical energy to light, the number of photons of yellow light it emits per second is
Options
A.3 × 1019
B.1.5 × 1020
C.6 × 1018
D.62 × 1020
Solution
Energy emitted = 200 × 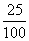 = 50 W = 50 J / sec.
= 50 W = 50 J / sec.
No. of photons =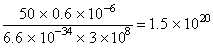
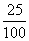 = 50 W = 50 J / sec.
= 50 W = 50 J / sec.No. of photons =
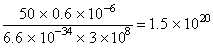
Create a free account to view solution
View Solution FreeMore Photoelectric Effect Questions
What wavelength is corresponding to a beam of electrons whose kinetic energy is 100eV (h = 6.6 × 10-34 J-S, 1eV = 1...The phenomenon of photoelectric effect was explained by :...What determines the hardness of the X-rays obtained from the Coolidge tube :-...The unit of luminous efficiency of an electric bulb is:...The energy of a photon of light with wavelength 5000 is approximately x eV. This way the energy of an X-ray photon with ...