s-Block ElementsHard
Question
One g equimolecular mixture of Na2CO3 and NaHCO3 is reacted with 0.1N HCl. The mL of 0.1 N HCl required to react completely with the above mixiture is :
Options
A.15.78mL
B.153.8mL
C.198.4mL
D.295.5mL
Solution
Na2CO3 + 2HCl → 2NaCl + H2O + CO2
NaHCO3HCl → NaCl + H2O + CO2
Mole of Na2CO3 =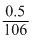 0.0047
0.0047
Mole of NaHCO3 =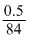 = 0.0059
= 0.0059
0.0047 mole of Na2CO3 ≡ 0.0047 × 2 mole of HCl
= 0.0094 mole of HCl 0.0059mole of 3 NaHCO3 ≡ 0.0059mole of HCl
Total mole required = 0.0059 moles of HCL If V be the volunes in litre of HCl required them mole of HCl required = V × 0.1
V =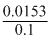 = 0.153 litre
= 0.153 litre
NaHCO3HCl → NaCl + H2O + CO2
Mole of Na2CO3 =
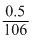 0.0047
0.0047Mole of NaHCO3 =
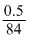 = 0.0059
= 0.00590.0047 mole of Na2CO3 ≡ 0.0047 × 2 mole of HCl
= 0.0094 mole of HCl 0.0059mole of 3 NaHCO3 ≡ 0.0059mole of HCl
Total mole required = 0.0059 moles of HCL If V be the volunes in litre of HCl required them mole of HCl required = V × 0.1
V =
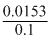 = 0.153 litre
= 0.153 litreCreate a free account to view solution
View Solution FreeMore s-Block Elements Questions
Which does not exist in solid state....Which of the following metal cation can be detected by flame test :...The molecular formula of plaster of pairs is :...Sodium carbonate can be manufactured by Solvay′s process but potassium carbonate cannot be prepared because:...Which alkali metal forms bicarbonate only in aqueous solution ?...