s-Block ElementsHard
Question
Sodium carbonate is soluble in water because of
Options
A.high lattice energy
B.low lattice energy
C.low molecular weight
D.high molecular weight
Solution
Sodium carbonate ionises in aqueous solution. It is solute in water because of low lattice energy. Low lattice energy helps in solubility. for substances which doe not ionise
Solubility ∝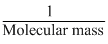
Solubility ∝
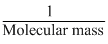
Create a free account to view solution
View Solution Free