NEETThermodynamicsHard
Question
Calculate change in internal energy if
ᐃH = - 92.2kJ, P = 40 atm and ᐃV = - 1L.
ᐃH = - 92.2kJ, P = 40 atm and ᐃV = - 1L.
Options
A.- 42 kJ
B.- 88 kJ
C.+ 88 kJ
D.+ 42 kJ
Solution
ᐃH = - 92.2kJ , P = 40atm, ᐃV = - 1L
Using ᐃH = ᐃE + PᐃV
⇒ ᐃE = ᐃH - PᐃV
=(- 92.2 kJ) - (40 atm) (-1L) ×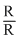
= 92.2kJ + (40 atm L) ×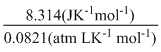
= - 92200J + 405.66J - 88149.34J
= - 88.149kJ ≈ - 88kJ
Using ᐃH = ᐃE + PᐃV
⇒ ᐃE = ᐃH - PᐃV
=(- 92.2 kJ) - (40 atm) (-1L) ×
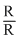
= 92.2kJ + (40 atm L) ×
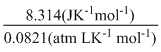
= - 92200J + 405.66J - 88149.34J
= - 88.149kJ ≈ - 88kJ
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
ΔS will be highest for which of the following reaction?...In which case will the efficiency of a Carnot cycle be higher, when the hot body temperature is increased by ΔT or when ...Two moles of an ideal gas (Cv,m = 1.5R) is subjected to the following changes in states.$A\left( \text{500 K, 5 bar} \ri...The increase of pressure on ice ⇋ water system at constant temperature will lead to :...Which of the following sets does show the intensive properties?...