ThermodynamicsHard
Question
One mole of an ideal gas for which Cv = (3/2) R is heated reversibly at a constant pressure of 1 atm from 25oC to 100oC. The ᐃH is :
Options
A.3.775 cal
B.37.256 cal
C.372.56 cal
D.3725.6 cal
Solution
ᐃH = 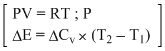
=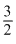 R × 75 + R × 75 = 75×
R × 75 + R × 75 = 75× 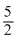
= 372.56 cal
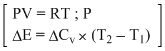
=
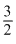 R × 75 + R × 75 = 75×
R × 75 + R × 75 = 75× 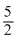
= 372.56 cal
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
Which of the following processes are expected to be spontaneous at low temperature and nonspontaneous at high temperatur...For which of the following process ΔS°sys is positive?...Select the correct option(s)....Which of the following is correct option for free expansion of an ideal gas under adiabatic condition ?...A piston filled with 0.04 mol of an ideal gas expands reversibly from 50.0 mL to 375 mL at a constant temperature of 37....