ThermodynamicsHard
Question
A gas expands isothermally against a constant external pressure of 1 atm from a volume of 10dm3 to a volume of 20dm3. It absorbs 300J of thermal energy from its surrounding. The ᐃU is :
Options
A.- 312 J
B.+123 J
C.- 213 J
D.+ 231 J
Solution
W = - 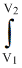 PdV = - P(V2 - V2)
PdV = - P(V2 - V2)
W = - (1atm) (20 - 10) = 10 dm3atm.
= - 10 dm3 ×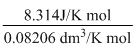 = - 1013 J
= - 1013 J
According to Ist law of thermodynamics, ᐃU = q + W = 800J + (-1013J) = - 213J
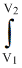 PdV = - P(V2 - V2)
PdV = - P(V2 - V2)W = - (1atm) (20 - 10) = 10 dm3atm.
= - 10 dm3 ×
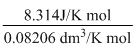 = - 1013 J
= - 1013 JAccording to Ist law of thermodynamics, ᐃU = q + W = 800J + (-1013J) = - 213J
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
For the reaction of the one mole of zinc dust with one mole of sulphuric acid in a bombcalorimeter, ᐃU and w corre...Among the following, the state function(s) is(are)...Which of the following is true for reversible adiabatic process involving an ideal gas?...At 300 K, the reactions which have the following values of thermodynamic parameters, occur spontaneously....One mole of ice is converted into water at 273 K and 1 atm. The entropies of H2O (s) and H2O (l) are 38.0 and 58.0 J/K-m...