Chemical BondingHard
Question
Select the correct statement
Options
A.If molecule has any polar bond then it is always polar
B.Solubility of noble gases decreases in water down the group when their size increases
C.London dispersion forces also contributes in net intraction between polar molecules
D.Energy of intraction between CF4 molecules is higher as compaired to molecular interaction between NF3 molecules
Solution
(A) For example: 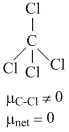
(B) Inert gas dissolves in water due to dipole - induced dipole not by London dispersion force

(B) Inert gas dissolves in water due to dipole - induced dipole not by London dispersion force
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
Which of the following is para-magnetic ?...Which of the following is least likely to behave as Lewis base ?...The hydrisation of central metal ion in the complex ion [CrCl2(NO2)2(NH3)2]- is :-...In which of the folloiwng species the interatomic bond angle is 109o28′ ?...Y and Z respectively are :-...