Fluid MechanicsHard
Question
If liquefied oxygen at 1 atmospheric pressure is heated from 50K to 300 K by supplying heat at constant constant rate. the graph of temperature vs time will be
Options
A.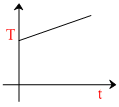

B.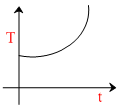

C.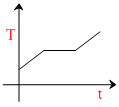

D.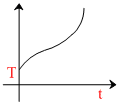

Solution
Q = mcᐃT
Q = mv(T - T0) ....(i)
Q = Kt whereas K is heating rate
∴ from 50 to boiling temprature, T increases linearly.
At vaporization, equation is Q = mL
so, temperature remains constant till vaporisation is complete
After that, again Eqn (i) is followedand temperature increase linearly
Q = mv(T - T0) ....(i)
Q = Kt whereas K is heating rate
∴ from 50 to boiling temprature, T increases linearly.
At vaporization, equation is Q = mL
so, temperature remains constant till vaporisation is complete
After that, again Eqn (i) is followedand temperature increase linearly
Create a free account to view solution
View Solution FreeMore Fluid Mechanics Questions
A cylindrical container of radius ′R′ and height ′h′ is completely filled with a liquid. Two hor...A weight is suspended from a long metal wire. If the wire suddenly breaks, its temperature...A ball falling in a lake of depth 200 m shows 0.1% decrease in its volume at the bottom. What is the bulk modulus of the...One end of a long iron chain of linear mass density λ is fixed to a sphere of mass m and specific density 1/3 while...A cylinder is filled with a liquid of density d upto a height h. If the beaker is at rest, then the mean pressure on the...