Carboxylic Acid and Acid DerivativesHard
Question
Which of the following is correct order of acidity?
Options
A.HCOOH > CH3COOH > ClCH2COOH > C2H5COOH
B.ClCH2COOH > HCOOH > CH3COOH > C2H5COOH
C.CH3COOH > HCOOH > ClCH3COOH > C2H5COOH
D.C2H5COOH > CH3COOH > HCOOH > ClCH2COOH
Solution
Recall that presence of electron-withdrawing group increases, while presence ofelectron-releasing group decreases the acidity of carbon Alic acids.
ClCH2COOH >
(electron-withdrawing gp)
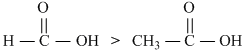
(Electron-releasing character increasing from Left to Right)
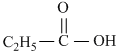
ClCH2COOH >
(electron-withdrawing gp)
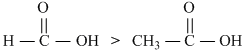
(Electron-releasing character increasing from Left to Right)
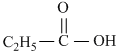
Create a free account to view solution
View Solution FreeTopic: Carboxylic Acid and Acid Derivatives·Practice all Carboxylic Acid and Acid Derivatives questions
More Carboxylic Acid and Acid Derivatives Questions
The treatment of an ester with LiAlH4 followed by acid hydrolysis produces :...Which of the following lacks chirality -...The compound which on reduction with LiAlH4 gives two alcohols -...Which of the following pair of compound would be easily hydrolysed. The pair contains one member from the two sets (1 & ...Hydrolysis of alkyl isocyanide yields :...