Hydrogen and its compoundHard
Question
12g of urea is dissolved in 1 litre of water and 68.4g of sucrose is dissolved in 1line of water. The lowering of vapour pressure of first case is
Options
A.equal to second
B.greater than second
C.less than second
D.double that of second
Solution
Moles of urea = 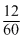 = 0.2
= 0.2
Moles of sucrise =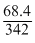 = 0.2
= 0.2
Both are non electrolyte hence lowering of V.P will be same.
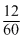 = 0.2
= 0.2Moles of sucrise =
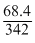 = 0.2
= 0.2 Both are non electrolyte hence lowering of V.P will be same.
Create a free account to view solution
View Solution FreeMore Hydrogen and its compound Questions
In the following reaction,Z is indentified as CH ≡ CH CH3CHO...The number of σ and π bonds present in ethane is :...Which compound does not dissolve in hot, dilute HNO3 ?...Which of the following species participatein sulphonation of benzene ring ?...At 0oC and 1 atm ice melt into water then the heat capacity of system is :-...