Redox and Equivalent ConceptHard
Question
An aqueous solution of 6.3 g of oxalic acid diihydrate is made up to 250 ml. The volume of 0.1 N NaOH required to completely neutralize 10 ml of this solution is
Options
A.20 ml
B.40 ml
C.10 ml
D.4 ml
Solution
Normalityof oxalic acid
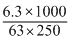 = 0.4 N
= 0.4 N
N1V1 = N2V2
10 × 0.4 = V × 0.1 = 40 ml
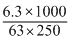 = 0.4 N
= 0.4 NN1V1 = N2V2
10 × 0.4 = V × 0.1 = 40 ml
Create a free account to view solution
View Solution FreeMore Redox and Equivalent Concept Questions
For the reaction I- + CIO3- + H2SO4 → Cl- + HSO4- + I2 The correct statements(s) in the balanced equation is/are...Which of the following ion cannot act as an oxidizing agent?...The oxide of a metal contains 52.91% of the metal. If the formula of the metal oxide is M2O3, then what is the atomic ma...Column - I Column-IIRedox Process n factor for underlined(a) As2S3 → AsO4- + SO4-2 (p) 4/3(b) I2 → I- + IO3-...An effective atomic number of [Co(CO)4] is 35 and hence is less stable. It attains stability by:-...