Chemical BondingHard
Question
A compound of a metal ion Mx+ (Z = 24) has a spin only magnetic moment of 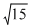 Bhor magnetons. The number of unparired electrons in the compound are
Bhor magnetons. The number of unparired electrons in the compound are
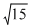 Bhor magnetons. The number of unparired electrons in the compound are
Bhor magnetons. The number of unparired electrons in the compound are Options
A.2
B.4
C.5
D.3
Solution
Magnetic moment μ = 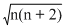
where n = number of unipaired electrons
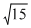 =
= 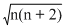
∴ n = 3
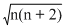
where n = number of unipaired electrons
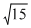 =
= 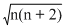
∴ n = 3
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
In the clathraates of xeonon with quinol, the nature of bonding between xenon and water molecule is....The ease of adsorption of the hydrated alkali metal ions on an ion-exchange resins follows the order:...The charge/size ratio of a cation determines its polarizing power. Which one of the following sequences represents the i...Among the following the maximum covalent character is shown by the compound :...Y and Z respectively are :-...