Hydrogen and its compoundHard
Question
The weight of NaCl decomposed by4.9g of H2SO4, if 6g of sodium hydrogen sulphate and 1.825 g of NCl, were produced in the reaction is :
Options
A.6.921 g
B.4.65 g
C.2.925 g
D.1.4 g
Solution
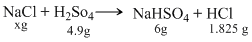
According to low of conservation of mass ″mass is neither created nor destroyed during a chemical change″
∴ Mass of the reactants = Mass of products
x = 4.9 = 6 + 1.825
x = 2.925 g
Create a free account to view solution
View Solution FreeMore Hydrogen and its compound Questions
At 0oC and 1 atm ice melt into water then the heat capacity of system is :-...Several blocks of magnesium are fixed to the bottom of a ship to...When hard water is passed through ion-exchange resin with formula RNH3+ OH-. It becomes free form :-...B.H.C. is used as an:...Given :-The enthalpy of the hydrogenation of these compounds will be in the order as :-...