ElectrostaticsHard
Question
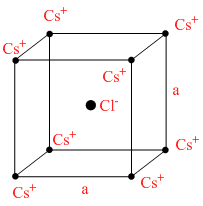
In the basic CsCl crystal structure, Cs+ and Cl+ ions are arranged in a bcc configuration as shown in the figure. The net electrostatic force exerted by the eight Cs+ ions on the ion is :

Options
A.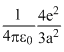
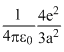
B.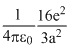
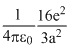
C.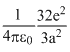
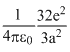
D.zero
Solution
All Cs ions are symmetrically distributed around Cl- so, resultant of all the fores acting on Cl- will be zero
Create a free account to view solution
View Solution FreeMore Electrostatics Questions
The electric potential varies in space according to the relation V = 3x + 4y. A particle of mass 0.1 kg starts from rest...The electric potential and field at a point due to an electric dipole are proportional to...The equation of equipotential line in an electric field is y = 2x, then the electric field strength vector at (1, 2) may...A unit positive point charge of mass m is projected with a velocity v inside the tunnel as shown. The tunnel has been ma...Let E be the electric field and V, the electric potential at a point....