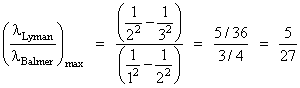NEET | 2013Atomic StructureHard
Question
Ratio of longest wavelengths corresponding to Lyman and Balmer series in hydrogen spectrum is:-
Options
A.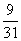
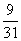
B.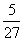
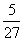
C.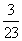
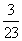
D.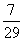
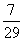
More Atomic Structure Questions
When 3Li7 nuclei are bombarded by protons, and the resultant nuclei are 4Be8, the emitted particles will be...Which of the following statements is correct?...Which on of the following is a possible nuclear reaction...In hydrogen spectrum the wavelength of Hα line is 656 nm; whereas in the spectrum of a distance galaxy Hα line...Holes are charged carries in...