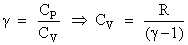NEET | 2013ThermodynamicsHard
Question
The molar specific heats of an ideal gas at constant pressure and volume are denoted by CP and CV, respectively. If 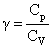 and R is the universal gas constant, then CV is equal to :
and R is the universal gas constant, then CV is equal to :
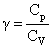 and R is the universal gas constant, then CV is equal to :
and R is the universal gas constant, then CV is equal to :Options
A.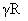
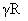
B.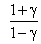
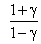
C.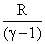
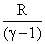
D.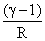
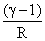
More Thermodynamics Questions
The specific heat of a gas at constant pressure is more than that of the same gas at constant volume because :-...Cp - Cv = R correctly applicable to which of the following :-...Which one of the following is not thermodynamical co-ordinate ?...A system is taken from its initial state (P1, V1) to a state (P2, V2). If the heat given to the system is ᐃQ and t...The correct value of 0oC on kelvin scale is :...