Heat TransferHard
Question
Cv and Cp denote the molar specific heat capacities of a gas at constant volume and constant pressure, respectively. Then
Options
A.Cp - Cv is larger for a diatomic ideal gas than for a monoatomic ideal gas.
B.Cp + Cv is larger for a diatomic ideal gas than for a monoatomic ideal gas.
C.Cp / Cv is larger for a diatomic ideal gas than for a monoatomic ideal gas.
D.Cp . Cv is larger for a diatomic ideal gas than for a monoatomic ideal gas.
Solution
For Monoatomic gas
Cv =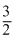 R, Cp =
R, Cp = 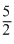 R
R
For diatomic gas
Cp =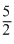 R Cp =
R Cp = 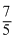 R
R
Cv =
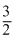 R, Cp =
R, Cp = 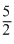 R
RFor diatomic gas
Cp =
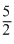 R Cp =
R Cp = 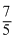 R
RCreate a free account to view solution
View Solution FreeMore Heat Transfer Questions
The temperature of the two outer surfaces of a composite slab, consisting of two materials having coefficients of therma...Two Plates of equal areas are placed in contact with each other. their thickness are 2 and 3 respectively. Temp of exter...Ice starts forming in lake with water at 0oC and when the atmospheric temperature is −10oC. If the time taken for ...Pyrometer is used in :...A heat flux of 4000 J/s is to be passed through a copper rod of length 10 cm and area of cross section 100 cm2. The ther...