MetallurgyHard
Question
The metal extracted by leaching with a cyanide is
Options
A.Mg
B.Ag
C.Cu
D.Na
Solution
Silver ore forms a soluble complex with NaCN from which silver is precipitated using scrap zinc.
Ag2S + 2NaCN → Na[Ag(CN)2]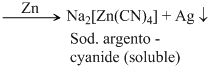
Ag2S + 2NaCN → Na[Ag(CN)2]
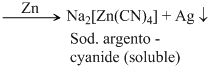
Create a free account to view solution
View Solution FreeMore Metallurgy Questions
In the leaching of Ag2S with NaCN, a stream of air is also passed. It is because of :...Carbon and CO gas are used to reduce which of the following pairs of metal oxides forexpiration of metals ?...Which of the following metals may be present in the anode mud during electrorefining of copper? I. Gold ; II. Iron, III....In froth-floatation process, pine oil functions as...Which of the following processes is used in extractive metallurgy of magnesium ?...