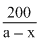Chemical Kinetics and Nuclear ChemistryHard
Question
The half - life of a radioisotope is four hours. If the initial mass of the isotope was 200 g, the mass remaining after 24 hours undecayed is
Options
A.1.042 g
B.4.167 g
C.3.125 g
D.2.084 g
Topic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
The following data certain to reaction between A and B ; Which of the following inference(s) can be drawn from the above...Which of the following is incorrect about freundlich adsorption isotherm ?...β - particle is emitted in radioactivity by...A.G.M. counter is used to study the radioactive process of first-order. In absence of radioactive substance A, it counts...Rate of a reaction can be expressed by Arrhenius equation as: k = A e-E /RT In this equation, E represents...