ThermodynamicsHard
Question
(ᐃH - ᐃU) for the formation of carbon monoxide (CO) from its elements at 298 K is (R = 8.314 J K-1 mol-1)
Options
A.- 1238.78 J mol-1
B.1238.78 J mol-1
C.- 2477.57 J mol-1
D.2477.57 J mol-1
Solution
ᐃH - ᐃU = ᐃngRT
= -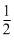 × 8.314 × 298
× 8.314 × 298
= - 1238.78
= -
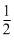 × 8.314 × 298
× 8.314 × 298= - 1238.78
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
The dissolution of NH4Cl in water is endothermic even through NH4Cl dissolves in water spontaneously. Which one of the f...A liquid is adiabatically expanded from state – I to state – II, suddenly by a single step, as shown in the figure then...Temperature of one mole of an ideal gas is increased by one degree at constant pressure. Work done by the gas is...Identify the intensive quantites from the following :...When a substance is heated, its entropy increases. The increase will be maximum at...