Chemical EquilibriumHard
Question
Phosphorus pentachloride dissociates as follows, in a closed reaction vessel,
PCl5(g) ⇋ PCl3(g) + Cl2(g)
If total pressure at equilibrium of the reaction mixture is P and degree of dissociation of PCl5 is x, the partial pressure of PCl3 will be
PCl5(g) ⇋ PCl3(g) + Cl2(g)
If total pressure at equilibrium of the reaction mixture is P and degree of dissociation of PCl5 is x, the partial pressure of PCl3 will be
Options
A.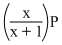
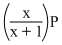
B.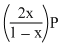
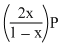
C.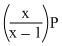
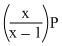
D.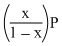
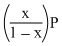
Solution
PCl5(g) ⇋ PCl3(g) + Cl2(g)
(1-x) x x
PPCl3 =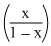 × P
× P
(1-x) x x
PPCl3 =
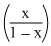 × P
× PCreate a free account to view solution
View Solution FreeMore Chemical Equilibrium Questions
The equilibrium $\text{SO}_2\text{Cl}_2(g) \rightleftharpoons \text{SO}_2(g) + \text{Cl}_2(g)$ is attained at $25^\circ\...Select incorrect reaction which is related to prepration of Cl2(g) -...NH4HS(S) ⇋ NH3(g) + H2S(g)The equilibrium pressure at 25oC is 0.660 atm. What is KP for the reaction....An amount of 3 moles of N2 and some H2 is introduced into an evacuated vessel. The reaction starts at t = 0 and equilibr...In a closed container maintained at 1 atm pressure and 25o C, 2 moles of SO2(g) and 1 mole of O2(g) were allowed to reac...