Atomic StructureHard
Question
Uncertainty in the position of an electron (mass = 9.1 × 10-31 kg) moving with a velocity 300 ms-1, accurate upto 0.001%, will be
Options
A.19.2 × 10-2 m
B.5.76 × 10-2 m
C.1.92 × 10-2 m
D.3.84 × 10-2 m
Solution
ᐃx. ᐃv ≥ 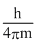
ᐃx ≥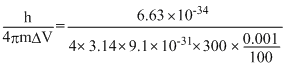
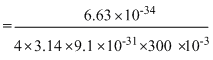
= 0.01933
= 1.93 × 10-2
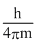
ᐃx ≥
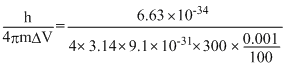
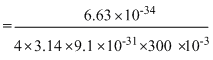
= 0.01933
= 1.93 × 10-2
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
dz2 - orbital has :...The wavelength associated with a golf ball weighing 200g and moving at a speed of 5 m / h is of the order :...From the α-particle scattering experiment, Rutherford concluded that...Select the correct expression(s) according to Bohr model -...The atomic nucleus contains :...