Chemical BondingHard
Question
Which of the following species exhibits the diamagnetic behaviour?
Options
A.O22-
B.O2+
C.O2
D.NO
Solution
The correct option is O22-
This species has 18 e-, which are filled in such a way that all molecular orbitals are fully filled, so diamagnetic.
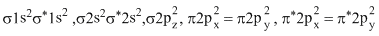
Hence, (1) is correct.
This species has 18 e-, which are filled in such a way that all molecular orbitals are fully filled, so diamagnetic.
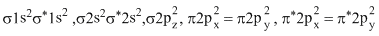
Hence, (1) is correct.
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
Arrange the following complex in increasing order of vibrational frequency of C - O bond -(I) [(PPh3)3Mo(CO)3] (II) [(Ph...Among the following groupings which represents the collection of isoelectronic species ?...The tendency of BF3, BCl3 and BBr3 to behave as Lewis acid decreases in the sequence...Reactant (A) and product (B) are respectively:...Match List-I with List-II according to shape.Choose the correct answer from the options given below :...