Periodic TableHard
Question
Which of the following atoms has the lowest ionization potential?
Options
A.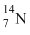
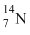
B.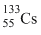
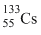
C.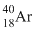
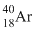
D.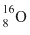
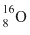
Solution
Since 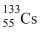 has larger size among the four atoms given, thus the electrons present in the outermost orbit will be away from the nucleus and the electrostatic force experienced by electrons due to nucleus will be minimum. Therefore the energy required to liberate electron from outer orbit will be minimum in the case of
has larger size among the four atoms given, thus the electrons present in the outermost orbit will be away from the nucleus and the electrostatic force experienced by electrons due to nucleus will be minimum. Therefore the energy required to liberate electron from outer orbit will be minimum in the case of 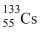
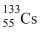 has larger size among the four atoms given, thus the electrons present in the outermost orbit will be away from the nucleus and the electrostatic force experienced by electrons due to nucleus will be minimum. Therefore the energy required to liberate electron from outer orbit will be minimum in the case of
has larger size among the four atoms given, thus the electrons present in the outermost orbit will be away from the nucleus and the electrostatic force experienced by electrons due to nucleus will be minimum. Therefore the energy required to liberate electron from outer orbit will be minimum in the case of 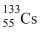
Create a free account to view solution
View Solution FreeMore Periodic Table Questions
In which of the following process energy is absorbed:...Select the correct statement :-...Which of the following is correctly matched :-...Which of the following is the correct statement :(a) Boron is diagonally related to silicion(b) Elements of third group ...Which one of the following sets of ions represents a collection of isoelectronic species?...