HydrocarbonsHard
Question
The hydrocarbon which can react with sodium in liquid ammonia is
Options
A.CH3CH2CH2C ≡ CCH2CH2CH3
B.CH3CH2C ≡ CH
C.CH3CH = CHCH3
D.CH3CH2C ≡ CCH2CH3
Solution
CH3CH2 - C ≡ CH 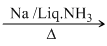 CH3CH2C ≡ -CNa+
CH3CH2C ≡ -CNa+
It is a terminal alkyne, having acidic hydrogen.
Note: Solve it as a case of terminal alkynes, otherwise all alkynes react with Na in liq. NH3.
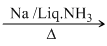 CH3CH2C ≡ -CNa+
CH3CH2C ≡ -CNa+It is a terminal alkyne, having acidic hydrogen.
Note: Solve it as a case of terminal alkynes, otherwise all alkynes react with Na in liq. NH3.
Create a free account to view solution
View Solution FreeMore Hydrocarbons Questions
Westrosol is a solvent & it is prepared by -...In the reaction CH3CH2CH2I + Zn−Cu + CH3CH2OH → the product expected is -...Marsh gas mainly contains :...Which of the following reactions does not involve a C−C bond formation −...Which of the following reactions will result in the formation of a chiral centre in the product -...