ThermodynamicsHard
Question
On the basis of the following thermochemical data : (áƒGfoH+(aq) = 0)
H2O(l) → H+ (aq) + OH-(aq); áƒH = 57.32 kj
H2(g) + 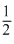 O2(g) → H2O(l) ; áƒH = - 286.20kJ
O2(g) → H2O(l) ; áƒH = - 286.20kJ
The value of enthalpy of formation of OH- ion at 25oC is :
Options
A.-22.88 kJ
B.-228.88 kJ
C.+228.88 kJ
D.-343.52 kJ
Solution
By adding the two given equations, we have
H2(g) + 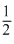 O2(g) → H+2(g) + OH-2(g) ; áƒH = -228.88 Kj
O2(g) → H+2(g) + OH-2(g) ; áƒH = -228.88 Kj
Here áƒHfo of H+(aq) = 0
∴ áƒHfo of OH+ = -228.88 kJ
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
Which one of the following characteristics is not correct for physical adsorption of a gas on solid?...In thermodynamics, a process is called reversible when...Which of the following statement(s) is/are correct regarding reversible adiabatic and isothermal processes for an ideal ...An ideal gas with the adiabatic exponent $\gamma$goes through a process P = Po – αV, where Po and α are positive constan...An ideal gas expands from volume V1 to V2. This may be achieved by any of the three processes, such as isobaric, isother...