NEET | 2018d and f-Block ElementsHard
Question
The geometry and magnetic behaviour of the complex [Ni(CO)4] are
Options
A.
square planar geometry and diamagnetic
B.
tetrahedral geometry and diamagnetic
C.
square planar geometry and paramagnetic
D.
tetrahedral geometry and paramagnetic
Solution
tetrahedral geometry and diamagnetic
Ni3d8 4s2
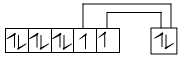
CO is SFL so unpaired electrons will get paired
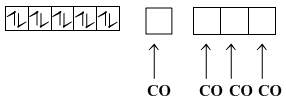
sp3 hybridisation
Tetrahedral, diamagnetic
Create a free account to view solution
View Solution Free