JEE Main | 2018Chemical BondingHard
Question
The predominant form of histamine present in human blood is (pKa, Histidine = 6.0)
Options
A.
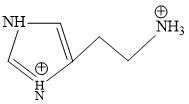
B.
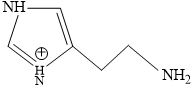
C.
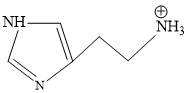
D.
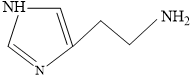
Solution
Structure of Histamine
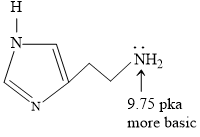
Blood is slightly basic in nature (7.35 pH) approx.
At this pH terminal NH2 will get protonated due to more basic nature.
Predominant structure of Histamine is
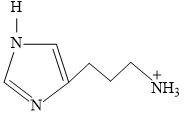
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
Among the following, the linear molecule is :...Which of the following structure is similar to graphite?...Which of the following lanthanoid ions is diamagnetic?(Atoms, Ce = 58, Sm = 62, Yb = 70)...(I) (II) (III) (IV)The correct order of decreasing basicity of the above compound :-...Which of the following is correct graph for the reaction ?...