JEE Main | 2014p-Block elementsHard
Question
Which one of the following properties is not shown by NO?
Options
A.It is diamagnetic in gaseous state
B.It is a neutral oxide
C.It combines with oxygen to form nitrogen dioxide
D.It′s bond order is 2.5
Solution
Nitric oxide is paramagnetic in the gaseous state as it has one unpaired electron in its outermost shell.
The electronic configuration of NO is
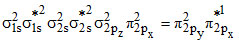
However, it dimerises at low temperature to become diamagnetic.
2NO ⇋ N2O2
Its bond order is 2.5 and it combines with O2 to give nitrogen dioxide.
The electronic configuration of NO is
However, it dimerises at low temperature to become diamagnetic.
2NO ⇋ N2O2
Its bond order is 2.5 and it combines with O2 to give nitrogen dioxide.
Create a free account to view solution
View Solution Free