General Organic ChemistryHard
Question
The correct order of decreasing acid strength of trichloroacetic acid (A), trifluoroacetic acid (B), acetic acid (C) and formic acid (D) is
Options
A.B > A > D > C
B.B > D > C > A
C.A > B > C > D
D.A > C > B > D
Solution
If an electron withdrawing group (I showing group) is present, it makes the removal of proton more easy by stabilising the remaning carboxylate ion and thus, makes the acid more acidic.
The order of acidity of given compounds is
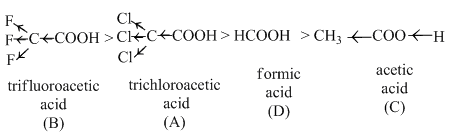
The order of acidity of given compounds is

Create a free account to view solution
View Solution FreeMore General Organic Chemistry Questions
Arrange basicity of the given compounds in decreasing order -(a) CH3 − CH2 − NH2 (b) CH2 = CH − NH2 (c...The order of stability of the following carbanion is : (I) (II) (III) (IV)...Which of the following shows the correct order of decreasing acidity -...Arrange the following compounds in order of decreasing acidity :...The correct stability order of following species is -...