Photoelectric EffectHard
Question
Two isotopes P and Q of atomic weight 10 and 20, respectively are mixed in equal amount by weight. After 20 days their weight ratio is found to be 1 : 4. Isotope P has a half-life of 10 days. The half-life of isotope Q is
Options
A.zero
B.5 days
C.20 days
D.inifinite
Solution
No. of nucleus of P, NP = 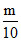 × NA
× NA
No. of nucleus of Q, NQ =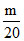
No. of Isotope P after 20 days, NP′ =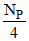
Let no. of Isotope Q after 20 days be NQ′
∴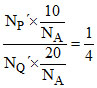 (given)
(given)
NQ′ = 2 × NP′ =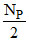 = NQ
= NQ
Thus no change in number of Q. Hence its half life is infinity.
No. of nucleus of Q, NQ =
No. of Isotope P after 20 days, NP′ =
Let no. of Isotope Q after 20 days be NQ′
∴
NQ′ = 2 × NP′ =
Thus no change in number of Q. Hence its half life is infinity.
Create a free account to view solution
View Solution FreeMore Photoelectric Effect Questions
Penetration power of X-rays depend on :...Which on of the following affects the alasticity of a substance ?...The wavelength of Kα-line characteristic X-rays emitted by an element is 0.32. The wavelength of Kβ-line emitt...X-ray falling on a material...-In Davisson - Germer experiment, Nickel crystal acts as :-...