Photoelectric EffectHard
Question
The atomic weight of boron is 10.81 g/mole and it has two isotopes 105B and 115B. The ratio (by number) of 105B : 115B in nature would be :
Options
A.19 : 81
B.10 : 11
C.15 : 16
D.81 : 19
Solution
Let 105B and 115B be in the ratio m:n .
Average atomic weight
10.81 =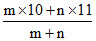
⇒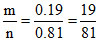
Average atomic weight
10.81 =
⇒
Create a free account to view solution
View Solution FreeMore Photoelectric Effect Questions
In a uranium reactor whose thermal power is P = 100 MW, if the average number of neutrons liberated in each nuclear spli...For first order X-ray diffraction if lattice constant is 3 × 10-8 cm and glancing angle is 30o, then the value of l...What determines the hardness of the X-rays obtained from the Coolidge tube :-...5000V is applied on an electronic X-ray tube. Then minimum wave length of X-ray will be -...The work done in splitting a drop of water of 1 mm radius in to 106 droplets is (surface tansion of water 72 × 10-3...