Photoelectric EffectHard
Question
When a hydrogen atom is excited from ground state to first excited state then
Options
A.its kinetic energy increases by 10.2 eV.
B.its kinetic energy decreases by 10 .2 eV.
C.its kinetic energy decreases by 10 .2 eV.
D.its angular momentum increases by 1.05 × 10-34 J-s.
Solution
K.E. = 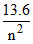 eV, P.E =
eV, P.E = 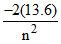 eV for Hydrogen z = 1
eV for Hydrogen z = 1
ᐃK = Kf − Ki z = 1
ᐃK.E. =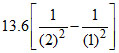 = − 10.2 eV (decrease)
= − 10.2 eV (decrease)
= (PE)f − (PE)i
ᐃP.E. =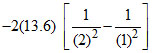 = 20.4 eV (increase)
= 20.4 eV (increase)
Angular momentum L =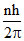
ᐃL =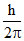 (2 − 1) =
(2 − 1) = 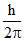 = 1.05 × 10-34 J-s (increase)
= 1.05 × 10-34 J-s (increase)
ᐃK = Kf − Ki z = 1
ᐃK.E. =
= (PE)f − (PE)i
ᐃP.E. =
Angular momentum L =
ᐃL =
Create a free account to view solution
View Solution FreeMore Photoelectric Effect Questions
Work of 3.0 × 10-4 joule is required to be done in increasing the size of a soap film from 10cm × 6cm to 10cm ...Bernulli equation is a consequence of conservation of :...Inverse-square law for illumination is valid for :...If h is the planck′s constant, the momentum of a photon of wavelength 0.01 is :...On reducing the intensity of light incident on a metal surface :...