Photoelectric EffectHard
Question
The different lines in the Lyman series have their wavelengths lying between :
Options
A.zero to infinit
B.900(Ao) to 1200 (Ao)
C.1000(Ao) to 1500 (Ao)
D.500(Ao) to 1000 (Ao)
Solution
for lyman series
shortest wavelength n = ∞ → n = 1
ᐃE = 13.6 eV
and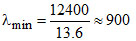 Å
Å
Longest wavelength n = 2 → n = 1
ᐃE = 10.2 eV
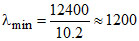 Å
Å
shortest wavelength n = ∞ → n = 1
ᐃE = 13.6 eV
and
Longest wavelength n = 2 → n = 1
ᐃE = 10.2 eV
Create a free account to view solution
View Solution FreeMore Photoelectric Effect Questions
Light of frequency v is incident on a metal of threshold frequency v0. Then work function of metal will be :-...In the Davisson and Germer experiment, the velocity of electrons emitted from the electron gun can be increased by...The energy that should be added to an electron to reduce its De broglie wavelength from 10-10m to 0.5 × 10-10 m wil...A sphere of mass M and radius R is falling viscous fluid. The terminal velocity attained by the falling object will be p...When X-rays tube is operated at V0 voltage, it produces Kα X-ray of wavelength l. Now voltage is made then waveleng...