Photoelectric EffectHard
Question
According to Bohr′s theory of the hydrogen atom, the speed vn of the electron in a stationary orbit is related to the principal quantum number n as (C is a constant) :
Options
A.vn = C/n2
B.vn = C/n
C.vn = C × n
D.vn = C × n2
Solution
Velocity of an electron is given by
V =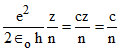
For H -atom (z = 1)
V =
For H -atom (z = 1)
Create a free account to view solution
View Solution FreeMore Photoelectric Effect Questions
Protons and singly ionized atoms of U235 & U238 are passed in turn (which means one after the other and not at the same ...What determines the hardness of the X-rays obtained from the Coolidge tube :-...The shortest wave length emitted from an X-ray tube depends upon...Shear modulus is zero for...92U235 nucleus absorbs a slow neutron and undergoes fission into 54X139 and 38Sr94 nuclei. The other particles produced ...