Photoelectric EffectHard
Question
In the Bohr model of the hydrogen atom, the ratio of the kinetic energy to the total energy of the electron in a quantum state n is :
Options
A.− 1
B.+ 1
C.1/n
D.1/n2
Solution
According to the Bohr model
P.E. = − 2 K.E. = 2 T. E.
⇒ K.E. = − T.E.
Where T.E. =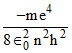
K.E. = −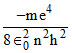 ⇒
⇒ 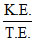 = −
= −
P.E. = − 2 K.E. = 2 T. E.
⇒ K.E. = − T.E.
Where T.E. =
K.E. = −
Create a free account to view solution
View Solution FreeMore Photoelectric Effect Questions
The solution of Bragg′s equation is possible if :...When X-rays tube is operated at V0 voltage, it produces Kα X-ray of wavelength λ. Now voltage is made then wav...Which of the following is true for photon :-...The energy of characteristic X-rays photon obtained from coolidge tube comes from :-...Which of the following is the wave length of X-ray:...