Photoelectric EffectHard
Question
If an orbital electron of the hydrogen atom jumps from the ground state to a higher energy state, its orbital speed reduces to half its initial value. If the radius of the electron orbit in the ground state is r, then the radius of the new orbit would be :
Options
A.2r
B.4r
C.8r
D.16r
Solution
Since speed reduces to half , KE reduced to
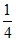 th ⇒ n = 2
th ⇒ n = 2
mvr =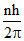
mv0 r = 1.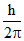 ..........I
..........I
m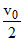 r′= 2
r′= 2 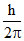 ..........II
..........II
r′ = 4.r
mvr =
mv0 r = 1.
m
r′ = 4.r
Create a free account to view solution
View Solution FreeMore Photoelectric Effect Questions
The bulk modulus of a metal is 1010 N/ m2 and Poisson′s ratio 0.20 If average distance between the molecules is 3A...What is the minimum wavelength of X-rays...Two stars situated at distances of 1 and 10 light years respectively from the earth appear to possess the same brightnes...When cathode rays strike a metal target of high melting point with very high velocity, then :-...Two lamps of luminous intensity of 8 Cd and 32 Cd respectively are lying at a distance of 1.2m from each other. Where sh...