Photoelectric EffectHard
Question
If an orbital electron of the hydrogen atom jumps from the ground state to a higher energy state, its orbital speed reduces to half its initial value. If the radius of the electron orbit in the ground state is r, then the radius of the new orbit would be :
Options
A.2r
B.4r
C.8r
D.16r
Solution
Since speed reduces to half , KE reduced to
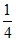 th ⇒ n = 2
th ⇒ n = 2
mvr =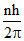
mv0 r = 1.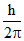 ..........I
..........I
m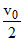 r′= 2
r′= 2 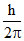 ..........II
..........II
r′ = 4.r
mvr =
mv0 r = 1.
m
r′ = 4.r
Create a free account to view solution
View Solution FreeMore Photoelectric Effect Questions
Which one of the following statements is NOT true for de Broglie waves ?...The wavelength of Kα-line characteristic X-rays emitted by an element is 0.32. The wavelength of Kβ-line emitt...Electron behaves like a wave because it -...If X-rays is passed through from strong magnetic field, then X-rays :-...Two separate monochromatic light beams A and B of the same intensity (energy per unit area per unit time) are falling no...