Photoelectric EffectHard
Question
Which energy state of doubly ionized lithium (Li++) has the same energy as that of the ground state of hydrogen ? Given Z for lithium = 3 :
Options
A.n = 1
B.n = 2
C.n = 3
D.n = 4
Solution
En (Li2+) = E1 (H)
⇒ − 13.6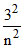 = −13.6 ×
= −13.6 × 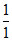
⇒ n = 3
⇒ − 13.6
⇒ n = 3
Create a free account to view solution
View Solution FreeMore Photoelectric Effect Questions
Water is filled in a container upto heigh of 3m. A small hole of area ′A0′ is punched in the wall of the con...A lead shot of 1mm diameter falls through a long column of glycerine. The variation of its velocity v with distance cove...Two photons having...Radiation pressure on any surface (for a given intensity):...The maximum kinetic energy of photoelectrons emitted from a surface when photons of energy 6 eV fall on it is 4 eV. The ...