Photoelectric EffectHard
Question
Consider 2 hydrogen like ions A and B. Ionization energy of A is greater than that of B. Let r, u, E and L represent the radius of the orbit, speed of the electron, energy of the atom and orbital angular momentum of the electron respectively. In ground state:
Options
A.rA > rB
B.uA > uB
C.EA > EB
D.LA > LB
Solution
ᐃE = ionization energy
= 13.6 × z2 eV
ᐃEA > ᐃEB ⇒ ZA > ZB
L =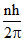
∴ LA = LB
E = energy of the orbit
EA < EB
u α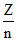
∴ uA > uB
r α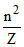
∴ rA < rB .
= 13.6 × z2 eV
ᐃEA > ᐃEB ⇒ ZA > ZB
L =
∴ LA = LB
E = energy of the orbit
EA < EB
u α
∴ uA > uB
r α
∴ rA < rB .
Create a free account to view solution
View Solution FreeMore Photoelectric Effect Questions
According to Mosley′s law, the frequency of a characteristic spectral line in X-ray spectrum varies as :-...The momentum of a photon in an X-ray beam of 10-10 metre wavelength is :-...Kα characteristic X-ray refers to the transition :-...The intensity of X-rays of wavelength 0.5 reduces to one fourth on passing through 3.5 mm. thickness of a metal foil. Th...Binding Energy per nucleon of a fixed nucleus XA is 6 MeV. It absorbs a neutron moving with KE = 2 MeV, and converts int...