KTGHard
Question
A fixed mass of an ideal gas undergoes changes of pressure and volume starting at L, as shown in Figure.
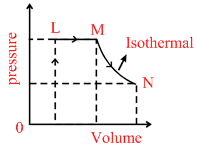
Which of the following is correct :

Which of the following is correct :
Options
A.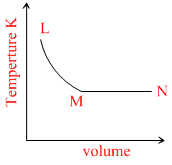

B.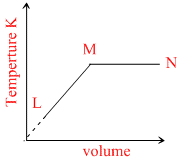

C.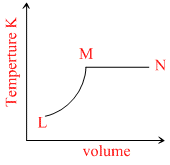

D.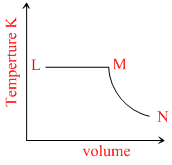

Solution
L → M P = constant V α T. MN T = constant
Here, option B is constant
Here, option B is constant
Create a free account to view solution
View Solution FreeMore KTG Questions
A piece of iron is heated in a flame. It first becomes dull red then becomes reddish yellow and finally turns to white h...At 27o C temperature, the kinetic energy of an ideal gas is E1. If the temperature is increased to 327o C, then kinetic ...If the total number of H2 molecules is double that of the O2 molecules then the ratio of total kinetic energies of H2 to...Two samples 1 and 2 are initially kept in the same state. The sample 1 is expanded through an isothermal process where a...For an ideal gas, the heat capacity at constant pressure is larger than than that at constant volume because...