KTGHard
Question
The process ᐃU = 0, for an ideal gas can be best represented in the form of a graph :
Options
A.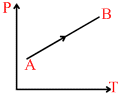

B.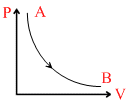

C.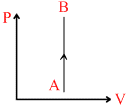

D.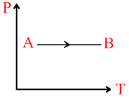

Solution
ᐃU = 0 ∴ T = constant
or PV = constant or P-V curve is a rectangular hyperbola.
clearly, option B is correct.
or PV = constant or P-V curve is a rectangular hyperbola.
clearly, option B is correct.
Create a free account to view solution
View Solution FreeMore KTG Questions
An ideal monoatomic gas undergoes the process AB as shown in the figure. If the heat supplied and the work done in the p...If the intermolecular forces vanish away, the volume occupied by the molecules contained in 4.5 kg. water at standard te...250 litre of an ideal gas is heated at constant pressure from 27o C such that its volume becomes 500 liters. The final t...For small coefficient of expansion in case of solid,...Avogadro′s number is the number of molecules :...