KTGHard
Question
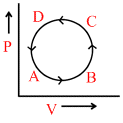
In the following figures (1) to (4), variation of volume by change of pressure is shown. A gas is taken along the path ABCDA. The change in internal energy of the gas will be:
(1)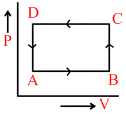 (2)
(2)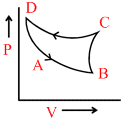 (3)
(3)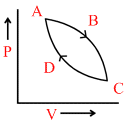 (4)
(4) 
(1)
 (2)
(2) (3)
(3) (4)
(4) 
Options
A.positive in all cases from (1) to (4)
B.positive in cases (1), (2) and (3) but zero in case (4)
C.negative in cases (1), (2) and (3) but zero in case (4)
D.zero in all the four cases.
Solution
As ᐃU = nRᐃT For closed path
ᐃT = 0 ∴ ᐃU = 0.
ᐃT = 0 ∴ ᐃU = 0.
Create a free account to view solution
View Solution FreeMore KTG Questions
When temperature is increased from 0oC to 273oC, in what ratio the average kinetic energy of molecules change ?...The pressure exerted by a gas in P0. If the mass of molecules becomes half and their velocities become double the pressu...Following is not a postulate of kinetic theory of gases :-...At temperature T, N molecules of gas A each having mass m and at the same temperature 2 N molecules of gas B each having...The adiabatic bulk modulus of hydrogen gas (γ = 1.4) at NTP is :...