ElectrostaticsHard
Question
The Gibbs energy for the decomposition of Al2O3 at 500oC is as follows:
2/3 Al2O3 → 4/3 Al + O2 ; ᐃrG = + 960kJ mol -1
The potential difference needed for the electrolytic reduction of aluminium oxide (Al2O3) at 500oCat least
2/3 Al2O3 → 4/3 Al + O2 ; ᐃrG = + 960kJ mol -1
The potential difference needed for the electrolytic reduction of aluminium oxide (Al2O3) at 500oCat least
Options
A.5.0 V
B.4.5 V
C.3.0 V
D.2.5 V
Solution
2/3 Al2O3 → 4/3 Al + O2
Al2O6 → 2Al3+ + 3O2-
ᐃG = - nFE ⇒ 960 × 103 = - 6 × 96500 × E
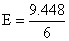 = 1.65 V required
= 1.65 V required
Al2O6 → 2Al3+ + 3O2-
ᐃG = - nFE ⇒ 960 × 103 = - 6 × 96500 × E
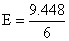 = 1.65 V required
= 1.65 V requiredCreate a free account to view solution
View Solution FreeMore Electrostatics Questions
A square surface of side L meter in the plane of the paper is placed in a uniform electric field E (volt/m) acting along...A uniformly charged thin spherical shell of radius R carries uniform surface charge density of σ per unit area. It...Choose the correct statements :-(a) The tangent drawn at any point on the line of force gives the direction of the force...A charge ′q′ is placed at the centre of a conducting spherical shell of radius R, which is given a charge Q....Let there be a spherically symmetric charge distribution with charge density varying as ρ (r) = ρ0 upto r = R,...