p-Block elementsHard
Question
When Cl2 gas reacts with hot and concentrated sodium hydroxide solution, the oxidation number of chlorine changes from
Options
A.zero to +1 and zero to -5
B. zero to -1 and zero to +5
C.zero to -1 and zero to +3
D.zero to +1 and zero to -3
Solution
When chlorine gas reacts with hot and concentrated NaOH solution, it disproportionates into chloride (Cl- ) and chlorate (Cl3-) ions.
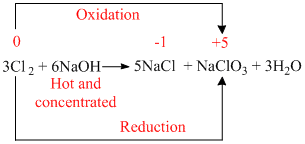
In this process, oxidation number of chorine changes from 0 to -1 and 0 to +5.
Note: In disproportionation reactions, the same element undergoes oxidation as well as reduction

In this process, oxidation number of chorine changes from 0 to -1 and 0 to +5.
Note: In disproportionation reactions, the same element undergoes oxidation as well as reduction
Create a free account to view solution
View Solution FreeMore p-Block elements Questions
Which of the following is least reactive ?...Peroxoborate in aqueous solution provides :...The reaction between ferric salt and aqueous sodium thiosulphate produces pink colour which soon diminishes. The pink co...Sulphur trioxide can be obtained by which of the following reaction...Inert gases are mixed in iodine vapours. Than there are between them....