Chemistry miscellaneousHard
Question
The % yield of ammonia as a function of time in the reaction
N2(g) + 3H2 (g) ⇋ 2NH3(g), ᐃH < 0
at (P, T) is given below -
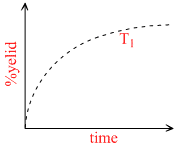
If this reaction is conducted at (P ,T2), with T2 > T1 , the % yield of ammonia as a function of time is represented by -
N2(g) + 3H2 (g) ⇋ 2NH3(g), ᐃH < 0
at (P, T) is given below -

If this reaction is conducted at (P ,T2), with T2 > T1 , the % yield of ammonia as a function of time is represented by -
Options
A.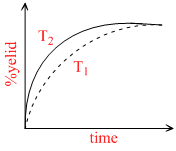

B.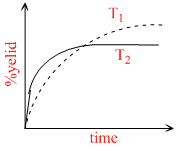

C.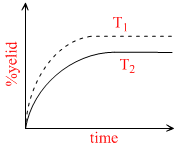

D.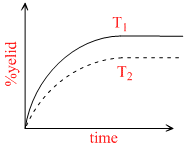

Solution
At t = 0 ⇒ rnet = kf [N2][H2]3 = rf
∴ & yield will increase in initial stages due to increase in net speed
As time proceeds ⇒ rnet = kf [N2][H2]3 - kb [NH3]2
On increasing temp., kf & kb increase but increase of kb is more so % yield will decrease
% yield will increase in initial stage due to enhance speed but as time proceeds , final yield is governed
by thermodynamics due to which yield decrease since reaction is exothermic
∴ & yield will increase in initial stages due to increase in net speed
As time proceeds ⇒ rnet = kf [N2][H2]3 - kb [NH3]2
On increasing temp., kf & kb increase but increase of kb is more so % yield will decrease
% yield will increase in initial stage due to enhance speed but as time proceeds , final yield is governed
by thermodynamics due to which yield decrease since reaction is exothermic
Create a free account to view solution
View Solution Free