Carboxylic Acid and Acid DerivativesHard
Question
Copper is purified by electrolytic refining of blister copper. The correct statement(s) about this process is (are)
Options
A.Impure Cu strip is used as cathode
B.Acidified aqueuous CuSO4 is used as electrolyte
C.Pure Cu deposits at cathode
D.Impurities settle as anode-mud
Solution
(A) Impure Cu strip is used as cathode : INCORRECT
Impure Cu strip is used as anode
(B) Aq.solution of (CuSO4 + H2SO4) is used as electrolyte : CORRECT
(C) Pure Cu deposites at cathode : CORRECT
Cu+2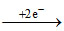 Cu
Cu
At cathode
(D) Impurities settle as anode mud : CORRECT
In electrorefining impurities are either soluble in electrolyte solution or deposit below anode known as anode mud.
Impure Cu strip is used as anode
(B) Aq.solution of (CuSO4 + H2SO4) is used as electrolyte : CORRECT
(C) Pure Cu deposites at cathode : CORRECT
Cu+2
At cathode
(D) Impurities settle as anode mud : CORRECT
In electrorefining impurities are either soluble in electrolyte solution or deposit below anode known as anode mud.
Create a free account to view solution
View Solution FreeTopic: Carboxylic Acid and Acid Derivatives·Practice all Carboxylic Acid and Acid Derivatives questions
More Carboxylic Acid and Acid Derivatives Questions
Which one of the following reactions can be used for the preparation of β-hydroxy ester :...The product A, B and C in the reaction sequenceABC are -...Which of the following acid radical gives ppt. with AgNO3 only under boiling condition and gives no ppt. under hot condi...Product obtained in above reaction will be -...Hofmann bromamide or hypobromite reaction is given by -amines...