JEE Advanced | 2015Atomic StructureHard
Question
One mole of a monoatomic real gas satisfied the equation p(V− b) = RT where b is a constant. The
relationship of interatomic potential V(r) and interatomic distance r for the gas is given by -
relationship of interatomic potential V(r) and interatomic distance r for the gas is given by -
Options
A.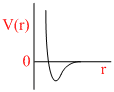

B.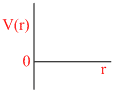

C.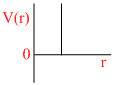

D.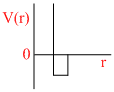

Solution
P (V- b) = RT
∵ a = 0
Since only repulsive forces are present.
Repulsive forces contribute only at very close distance.
So potential energy increases abruptly.
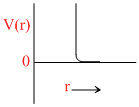
∵ a = 0
Since only repulsive forces are present.
Repulsive forces contribute only at very close distance.
So potential energy increases abruptly.

Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Which of the following relates to photons both as wave motion and as a stream of particles ?...The time period of revolution of electron in H-atom is directly proportional to...The orbital angular momentum of an electron is $\sqrt{3}\frac{h}{\pi}$. Which of the following may be the permissible va...In the Bohr’s atomic model, the electrostatic force of attraction between nuclear charge (Ze) and electron of charge e i...The radius of first orbit of H-atoms is 0.529 Å. The radius of first orbit of D-atoms should be...